How to Develop a Healthcare App
A Step-by-Step Guide
As an ISO 13485-certified provider, ScienceSoft has been delivering medical software that improves hospital workflows, care management, and patient experience since 2005.
App Development for Healthcare in Brief
Healthcare app development implies designing, architecting, coding, and supporting healthcare administration systems, patient apps, solutions for care management, and other clinical tools.
How to build a medical app in 7 steps
- Engineer functional and compliance requirements.
- Plan a compliance framework (HIPAA, GDPR, etc.) for sensitive data regulation within the SDLC.
- Design a secure architecture.
- Determine user personas and design an accessible UI.
- Build a minimum viable product (MVP).
- Get FDA approval (for medical devices or SaMD).
- Perform post-market monitoring and maintenance.
The development process may take from 4 months for an MVP of a simple mobile application (e.g., a patient-doctor communication or medication intake monitoring app) to 12+ months for a full-fledged solution like a remote patient monitoring system.
The costs of custom web or mobile healthcare software development may range from $40,000 to $2,000,000. The exact pricing will depend on software type, complexity, user roles, platform specifics, etc. Use our free calculator to estimate the cost for your case.
Explore the Diversity of Healthcare Software
Healthcare applications cater to the needs of healthcare organizations, medical professionals, and patients. Below are some of the solutions ScienceSoft delivers:
Health App Development Plan
The specifics of the medical software development process will depend on the app's purpose. This is a generalized plan elaborated by ScienceSoft's healthcare IT consultants based on our 20 years of experience in the field.
1.
Conceptualization and requirements engineering
This stage involves defining the software concept and analyzing the market and target audience (for software products). If you are a healthcare startup, you need to set your app apart from competitors to survive and attract investment. So, it would help if you found a niche related to a specific care type or patient group (e.g., easy-to-use apps for geriatric patients).
The next step is to identify the main features to be implemented. Common prioritizing methods, such as the Must, Should, Could, and Won’t (MoSCoW) method, KANO model, or value vs. effort, can help. The SRS should include non-functional requirements for security such as encryption, access controls, and audit trails.
At this stage, it’s also essential to define the regulatory requirements the software must comply with. If it will be used in the US, HIPAA compliance is a must as nearly all healthcare software deals with personal health information (PHI). Software for medical devices or SaMD additionally requires FDA approval.
2.
Project planning
The project plan outlines the budget, resource allocation, timelines, and deliverables. It’s important to allocate substantial time to testing and schedule regular compliance checks and code reviews. Prepare your team to maintain detailed documentation of the development process and create a risk mitigation plan.
3.
Architecture design
When designing the architecture, evaluate the potential options in terms of how well they meet the performance, scalability, security, and other requirements for the software. Most common evaluation approaches, like the architecture tradeoff analysis method (ATAM), might be effort-consuming. So, lightweight methods such as active reviews of intermediate designs (ARID) or pattern-based architecture review (PBAR) can be chosen. ARID, for example, can be used to ensure HIPAA compliance by identifying any potential privacy or security risks early on. With PBAR, the software architecture can be assessed for alignment with the healthcare industry standards.
Integrations are another essential part of architecture design. While this step depends on project specifics, healthcare apps are often integrated with EHR, CRM, PACS, LIS, billing systems, telemedicine platforms, and other software used by providers. For high software interoperability, uniform data exchange standards such as HL7, FHIR, and DICOM should be supported.
4.
UX and UI design
The UX/UI design must be developed to meet the needs of users, in accordance with their user roles (patients, medical staff, etc.) and corresponding user scenarios (e.g., patients can book an appointment or manage medications, doctors can access patients’ medical data, materials managers can order supplies).
It’s crucial to ensure usability for every user role. For example, for the patient interface, possible physical and cognitive restrictions should be considered to adapt the app to cope with them. This can be achieved by using high color contrast, large font sizes, text-to-speech, etc. Intuitive navigation can significantly enhance user engagement. Importantly, there should be easy-to-digest consent forms and a privacy policy for patients outlining data access and usage details to ensure regulatory compliance.
5.
Development and testing
It’s better to start with developing a minimum viable product (MVP), as it allows you to benefit from an early software launch and quickly adapt the software to user feedback.
Rigorous testing (often in parallel with development) is required to ensure the high quality of the software:
-
Performance testing helps ensure the app can handle stress conditions (e.g., an increased number of users).
-
Integration testing verifies that healthcare software modules or applications (e.g., EHR, patient apps, medical devices, imaging and lab software, etc.) work together smoothly.
-
Security testing is essential to guarantee data protection.
-
Functional testing confirms that the app works exactly as planned (e.g., prescriptions are filled correctly, insurance claims are processed, etc.).
-
Compliance testing verifies the software's adherence to regulatory standards such as HIPAA, IEC 62304, and FDA requirements.
-
Accessibility testing helps ensure usability for different user groups, including individuals with disabilities.
6.
Pre-launch activities and release
The last stage before the release requires revising the project documentation for consistency and conducting a HIPAA compliance assessment or GDPR compliance audit. In the case of software for medical devices or SaMD, you will also need to submit your product to FDA/MDR. Low-to-moderate-risk devices (e.g., software connected to a glucose monitor) only need premarket notification, while high-risk devices (e.g., medical diagnosis software) undergo premarket approval. Lastly, make sure to carry out the final review of security controls and draw up an incident response plan before launching the app.
7.
Post-launch activities
After releasing the app, it’s important to help the end users incorporate it into their routines. Successful adoption hinges on comprehensive user training and responsive software support, especially during the first few months after launch. It’s advisable to get user feedback to identify major concerns, which can be used to map out the app's next version. With time, software can also be updated to include new functionality. For example, AI can be used for intelligent diagnosing and treatment plan generation, as well as to enhance patient experience with the help of chatbots or virtual assistants.
Why Choose ScienceSoft for Web or Mobile Health App Development
- Healthcare app development company since 2005 in the domain.
- 150+ successful healthcare IT projects.
- ISO 13485, ISO 9001, and ISO 27001 certificates.
- Hands-on experience with HIPAA, GDPR, FDA, Cures Act, MDR, SAMHSA, CEHRT, and SAFER regulations.
- Proficiency in healthcare standards and terminologies such as HL7, FHIR, ICD-10, SNOMED CT, LOINC, RxNorm, CPT, XDS/XDS-I, DICOM, Blue Button+, X12 837/835, CDA, and CCD.
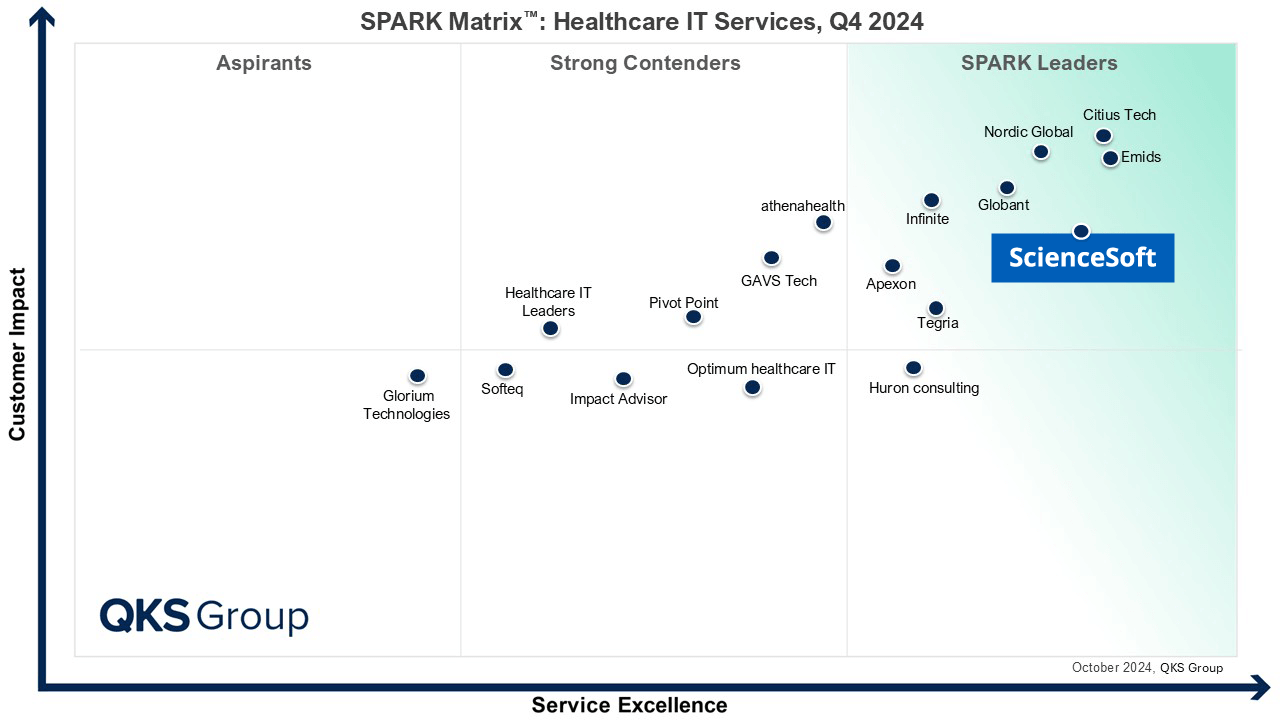
Featured among Healthcare IT Services Leaders in the 2022 and 2024 SPARK Matrix
Recognized for Healthcare Technology Leadership by Frost & Sullivan in 2023 and 2025
Named among America’s Fastest-Growing Companies by Financial Times, 5 years in a row

Top Healthcare IT Developer and Advisor by Black Book™ survey 2023
Four-time finalist across HTN Awards programs

Named to The Healthcare Technology Report’s Top 25 Healthcare Software Companies of 2025

HIMSS Gold member advancing digital healthcare
ISO 13485-certified quality management system
ISO 27001-certified security management system
Sourcing Models for Healthcare Software Development
Technologies ScienceSoft Is Experienced In
Low-code development
Clouds
DevOps
Containerization
Automation
CI/CD tools
Monitoring
Healthcare App Development Costs
Below are some software cost factors to be considered:
- Scope and software type (telemedicine, RPM, EHR, etc.).
- Platform (e.g., web, mobile, desktop).
-
Healthcare mobile app development approach (cross-platform vs. native iOS / Android healthcare app development).
- Number of user roles (patients, doctors, nurses, etc.) and the expected number of users.
- Integrations (e.g., with EHR, wearables, etc.).
- Regulatory compliance requirements (HIPAA, GDPR, FDA requirements, etc.).
- Performance, security, usability, and accessibility requirements.
- Required software maturity (an MVP or a fully-featured solution).
- Sourcing model (in-house vs. outsourcing).
The cost of custom healthcare app development varies from $40,000 for a simple mobile patient app (e.g., medication intake reminder) to $2,000,000+ for a complex EHR system with diverse functionality. Below are some sample cost ranges for popular software types:
From $40,000 to $300,000
for a patient app
From $200,000 to $400,000
for telemedicine software
From $200,000 to $400,000
for remote patient monitoring or medical device software
From $300,000 to $800,000
for a digital therapeutics solution
From $400,000 to $2,000,000+
for EHR software
Get a Cost Estimate for Your Healthcare Software Development Project
Please answer a few questions about your software development needs. This will help our team provide a tailored service offering and a cost estimate much quicker.
Thank you for your request!
We will analyze your case and get back to you within a business day to share a ballpark estimate.
In the meantime, would you like to learn more about ScienceSoft?
- Project success no matter what: learn how we make good on our mission.
- Since 2005 in healthcare IT services: check what we do.
- 4,200+ successful projects: explore our portfolio.
- 1,400+ incredible clients: read what they say.











