IT Infrastructure Services for Life Sciences Companies
ScienceSoft is an ISO 27001-certified technology partner with 21 years of experience in healthcare and life sciences IT. Our clients in the pharmaceutical, biotech, medtech, clinical research, and diagnostic fields get IT infrastructures that stay secure and compliant while adapting to growth, changing priorities, and regulatory shifts.
Life sciences IT infrastructure services are provisioning and managing hardware, software, and networking foundations of research, clinical, and manufacturing workflows, including validated and regulated systems.
ScienceSoft helps life sciences companies keep infrastructure secure, stable, and ready for change while protecting validated systems and GxP-relevant operations from unnecessary risk.
Clear governance and accountability
We work within your approval, change control, and validation processes. Our team follows clear responsibility boundaries and tailored SLAs, so infrastructure work never puts validated systems or GxP-relevant operations at risk.
Security by design
Our engineers build network segmentation, access controls, and encryption into every infrastructure layer and align these controls with ISO 27001, NIST CSF, and CIS Benchmarks.
Observable operations
Our teams monitor system health, track service levels, and catch performance and capacity issues early. We tune observability so your teams get useful signals, less alert noise, and better control over cloud spend.
Safer scaling and modernization
We automate infrastructure operations, establish risk-based change workflows, and build modular foundations that help life sciences organizations scale, modernize, and introduce change with less configuration drift and fewer unexpected side effects.
Who We Serve
Learn more about the industry-specific software we support and the full range of services we deliver for each sector.
End-to-End IT Infrastructure Services for Life Sciences Companies
Managed IT Infrastructure for Life Sciences Companies
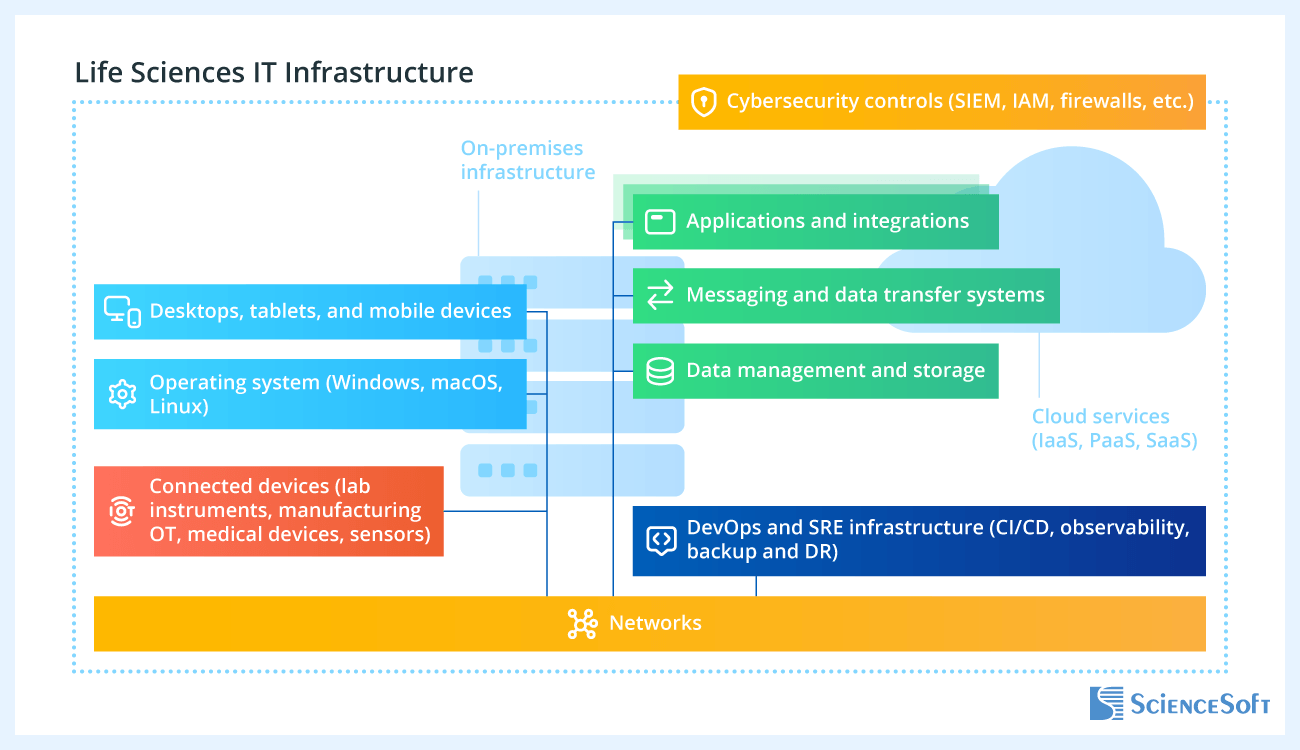
On-premises infrastructure
- Networks (LAN, WAN, Wi-Fi, SD-WAN, VPN).
- Compute platforms (servers, virtual machines, virtualization clusters).
- Storage, backup, and recovery.
Cloud and platform services
- Cloud infrastructure (IaaS, PaaS).
- Container platforms (Kubernetes, registries).
- Development platforms (CI/CD, development and test environments).
- Observability platforms (monitoring, logging, alerting).
Data platforms and integration
- Data platforms (databases, data warehouses, data lakes, lakehouses).
- Integration and interoperability (APIs, ETL/ELT, streaming, secure file transfer, application interfaces, monitoring).
Security and identity
- Identity and access management (SSO, MFA, privileged access).
- Network and endpoint security controls (NAC, firewalls, IDS/IPS, antimalware, EDR).
- Security monitoring and response tools (SIEM, SOAR, XDR).
- Data protection (DLP, data classification and labeling).
End-user and device landscape
- End-user computing (Windows, macOS, mobile, VDI).
- Laboratory and medical devices (lab instruments, IoT sensors, connected medical devices, telemetry, remote monitoring).
- Operational and manufacturing technology (manufacturing equipment, plant networks, process control, SCADA).
Applications and regulated operations
- Business, research, clinical, quality, and manufacturing systems (e.g., LIMS, CTMS/EDC, QMS, MES, SCM, imaging, portals).
- Regulated environments (GxP validation support, audit trails, data integrity controls).
Why Life Sciences Organizations Partner With ScienceSoft
- In cybersecurity since 2003.
- In healthcare and life sciences IT since 2005.
- In ITIL-compliant ITSM since 2008.
- In IT infrastructure services since 2010.
- A well-rounded team of architects, network engineers, system administrators, IT support agents, DevOps engineers, cybersecurity experts, and other talents. Over 50% of our IT professionals are senior-level experts.
- A portfolio of 150+ projects for healthcare and life sciences.
- Hands-on experience with GMP/GLP/GCP, FDA 21 CFR Part 11, and EU GMP Annex 11, as well as CSV and GAMP-based validation.
- Expertise in medical device quality management, including ISO 13485 and FDA 21 CFR Part 820, and IEC 62304 software lifecycle processes for regulated medical device software.
- Experience with data privacy regulations (HIPAA, GDPR) and interoperability requirements (21st Century Cures Act and ONC rules, where applicable) for PHI-related use cases.
- Proficiency in interoperability standards and datasets (FHIR, HL7, CCDA, USCDI) and clinical terminologies (SNOMED CT, LOINC, RxNorm, ICD-10, CPT).
- Ready to sign a BAA (Business Associate Agreement), NDA, or similar privacy agreements in case our engineers have access to systems that handle PHI.
Choose a Cooperation Model That Suits Your Needs
Consulting
We assess your infrastructure and work with your team to shape a solution that fits your environment and compliance needs. We deliver a practical roadmap with prioritized improvements and risk controls.
Partial outsourcing
You choose specific functions or tasks to hand over to ScienceSoft, such as cloud operations, network management, infrastructure automation, or security testing.
Full outsourcing
We manage the entire IT infrastructure, maintain and evolve its components, and continuously optimize cost and performance while supporting controlled change and evidence for regulated systems.
Cost Factors and Sample Estimates for Life Sciences IT Infrastructure Services
What affects the cost
Pricing depends on several factors, including:
- Service scope and delivery model: consulting, one-time implementation, modernization, or ongoing managed services, and which components are in scope (networks, cloud, data platforms, applications).
- Support coverage and workload: service hours (e.g., 24/7 vs. 8/5), support level (L1, L2, L3), and expected ticket volumes for support and maintenance.
- Validation and evidence scope for regulated and validated systems, including the depth of documentation, controlled change workflow, and audit-ready evidence required.
- Scale and complexity of the IT environment the infrastructure supports: number of users, hybrid versus cloud-only, number of systems and integrations, and change velocity.
- Security and compliance requirements: required standards or regulations, security controls, and security testing cadence.
Sample price ranges for managed services
These are sample ranges and meant as ballpark examples, not fixed prices.

- Infrastructure support (L3) cost is about $21,000 per month for a 500-employee company, $33,000 for 1,000 employees, and $67,000 for 2,000 employees.
- Application management and maintenance costs range from $3,000 per month for a stable app with minor change requests to $17,000+ per month for a large enterprise app in perpetual evolution.
Explore our dedicated page to learn how we ensure optimal pricing for each case.
Managed IT pricing models we rely on include:
- Subscription-based model.
- Fixed monthly fee.
- Per-ticket pricing.
- Time and materials (T&M).
Get a Tailored Estimate of Life Sciences IT Infrastructure Services
Use our cost configurator to get a free, non-binding ballpark estimate. Select the services you need, your preferred coverage, and compliance requirements, and we will share an initial estimate in 24 hours.
Thank you for your request!
We will analyze your case and get back to you within a business day to share a ballpark estimate.
In the meantime, would you like to learn more about ScienceSoft?
- 17 years in IT support: check what we do.
- 4,000 successful projects: explore our portfolio.
- 1,300+ incredible clients: read what they say.







